In the mesmerizing world of genetic engineering, prime editing has emerged as a shining star, promising to rewrite the very fabric of life with pinpoint precision. But here’s the plot twist: a team of relentless scientists at the Broad Institute of MIT and Harvard have summoned the powers of phages and protein engineering to birth a new generation of prime editors that are not just efficient but astonishingly compact.
The Genesis of Prime Editing
Prime editing, akin to the artistry of a genetic sculptor, was unveiled in 2019 by the visionary David Liu and his team. This editing technique entails genome modification through search-and-replace mechanisms, all achieved without the need for double-strand breaks or donor DNA. This molecular wizardry combines a tweaked Cas9 protein, an engineered prime editing guide RNA (pegRNA), and a modified reverse transcriptase enzyme. Together, they form a potent triumvirate capable of performing DNA edits with laser-like precision.

Epic Evolution: Phages to the Rescue
Enter the heroes of our tale: bacteriophages, the viruses that prey on bacteria. The scientists harnessed the evolutionary prowess of these tiny warriors through a technique called phage-assisted continuous evolution (PACE), first forged by David Liu in 2011. Picture this: a battlefield in the lab where only the bacteriophages that bear the most desirable genetic traits can survive and thrive, replicating faster than they can be wiped out.
In this grand showdown, the bacteriophages carrying the secrets of prime editing faced countless generations of evolution. The result? Brand new, turbocharged reverse transcriptases that turbocharge prime editing to unparalleled heights.
Prime Editors: Not All Heroes Wear Capes
Here’s the twist in our tale: Not all prime editors are cut from the same genetic cloth. The evolved prime editors have their own unique superpowers. Some excel in adding or swapping just a few DNA letters, while others stumble in these tasks but soar when it comes to inserting long DNA sequences. This revelation shattered the notion that one prime editor could rule them all.
Expanding the Editing Universe
With their newfound superpowers, the prime editors, now known as PE6a through PE6g, have blown the lid off previous limitations. These titans are two to 20 times more efficient than their predecessors, raising the banner of hope for therapeutic applications higher than ever before.
Their versatility shines bright as they effortlessly insert lengthy DNA sequences—ranging from 38 to 108 base pairs—into the genomes of human cells. Even the once-untamable frontier of live mouse brains has succumbed to their precision, with editing efficiency soaring to astonishing heights.
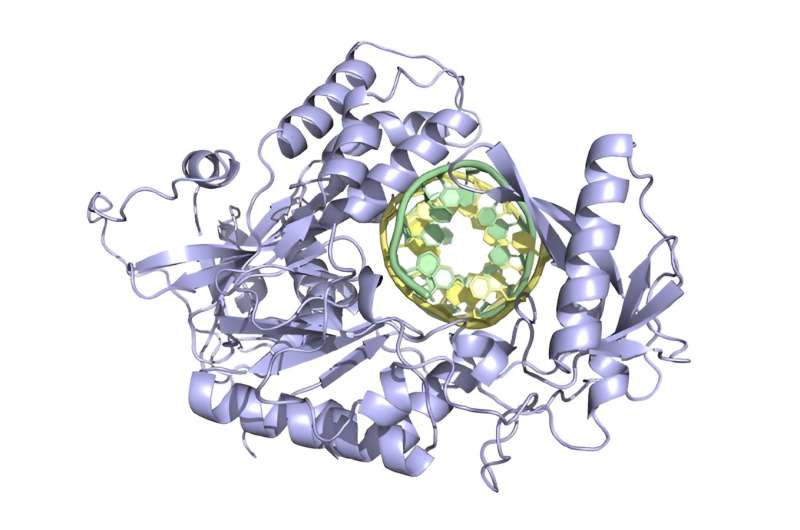
Prime Editors: Travel Light, Edit Right
But there’s more to this story. These newfound prime editors have slimmed down, shedding excess genetic baggage. This means they can squeeze into delivery systems that previously dared not accommodate them. This improved agility opens the door for prospective therapies in animal models as a necessary preliminary step before human studies.
You can find the complete article by following this link, and for additional updates on phage-related technologies and other related news, please explore our news section (The Phage News section).
Article reference: Jordan L. Doman et al, Phage-assisted evolution and protein engineering yield compact, efficient prime editors, Cell (2023). DOI: 10.1016/j.cell.2023.07.039

Leave a Reply
You must be logged in to post a comment.