Sewage is a rich source of diverse bacteriophages, particularly coliphages that target coliform bacteria. However, its composition can vary significantly depending on the source, such as residential, industrial, or hospital effluents, which may affect phage recovery. Like any other laboratory sample, sewage must be processed after collection to retain only the relevant portion for analysis. This guide provides a detailed method for sewage sample processing for bacteriophage isolation, highlighting key equipment, consumables, and handling considerations.
Step 1: Understanding Sewage Variability
- Residential Sewage: Typically contains organic waste, household chemicals, and a high load of coliform bacteria. Phage isolation is generally straightforward.
- Industrial Sewage: May contain heavy metals, chemicals, and lower bacterial content. Requires careful handling to avoid contamination and potential phage inactivation.
- Hospital Effluents: Rich in antibiotic-resistant bacteria and pharmaceuticals. Requires biosafety precautions due to potential pathogenic content.
Handling Tip: Always wear appropriate PPE (gloves, lab coat, eye protection) and handle samples in a biosafety cabinet when dealing with hazardous sewage sources.
Step 2: Required Equipment and Consumables
- Collection: Sterile sampling bottles (preferably 500 ml), icebox for transport.
- Preprocessing:
- Centrifuge (with high-speed capability, ~10,000 x g)
- Sterile filters (0.22 µm pore size for bacteria removal)
- Vacuum filtration system or syringe filters
- Sterile conical tubes (50 ml)
- Pipettes and sterile tips
- Vortex mixer
Step 3: Sample Preprocessing
- Initial Settling: Let the sewage sample sit at 4°C for 30 minutes to settle large debris.
- Decanting: Carefully pour the supernatant into a sterile tube, avoiding the disturbance of settled particles.
- Centrifugation: Spin the supernatant at 4,000 x g for 10-20 minutes to pellet the remaining debris.
- Filtration: Filter the supernatant through a 0.22 µm sterile filter to remove bacteria, retaining bacteriophages in the filtrate.
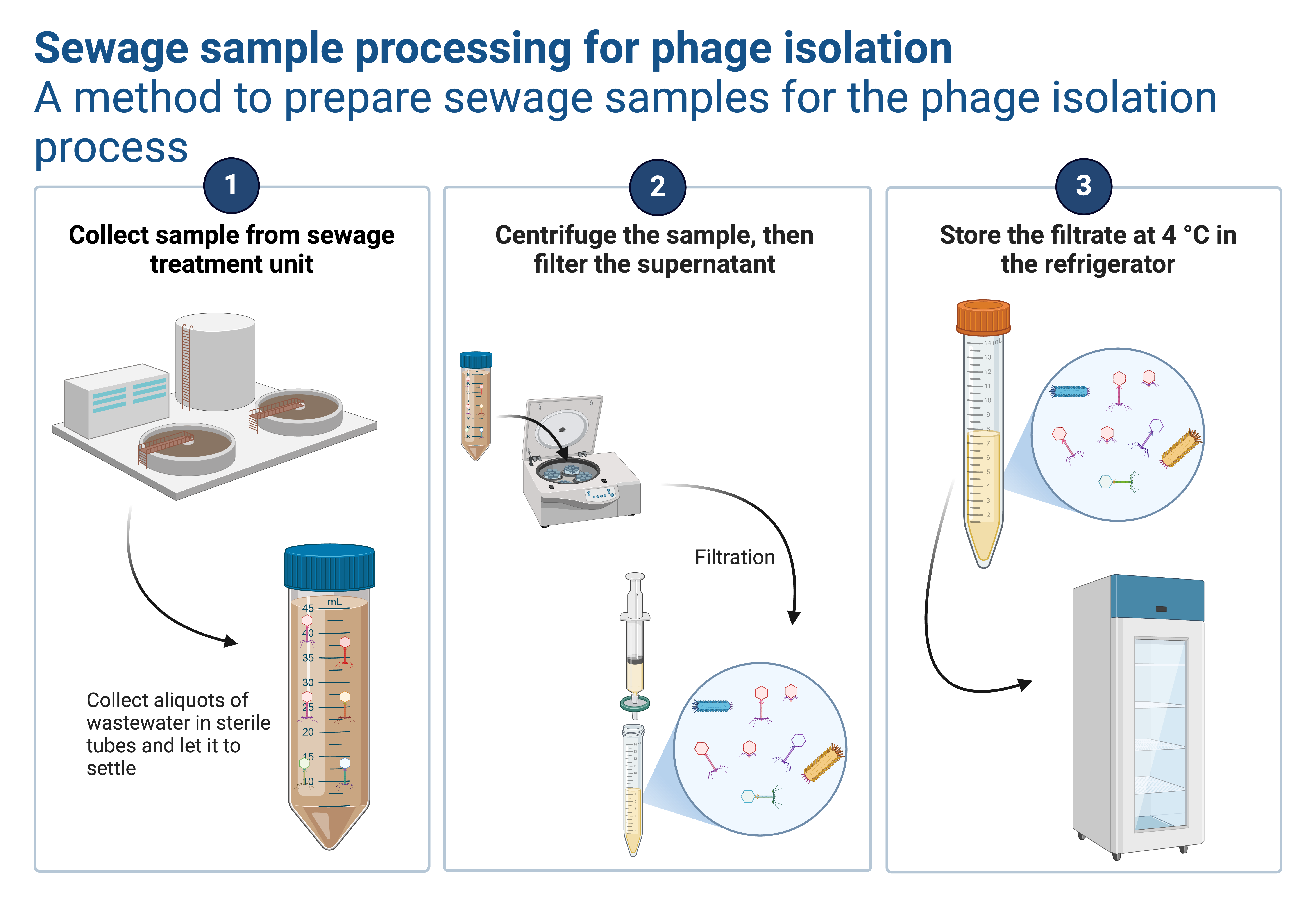
Handling Tip: For industrial or hospital sewage, consider an additional filtration step with a 0.45 µm filter before the 0.22 µm filter to prevent clogging.
Proper sewage sample processing is crucial for effective bacteriophage isolation. By understanding the source variability and using appropriate equipment and techniques, researchers can optimize phage recovery for downstream applications.
References
Image by The phage using Biorender

I do not even know how I ended up here, but I thought this post was great.
I don’t know who you are but certainly you are
going to a famous blogger if you are not already 😉 Cheers!