 |
| media preparation |
Follow these steps, and you’ll get great, or even perfect, agar plates – with no lumps, bubbles, or excess moisture – every time.
Some Tips for Pouring Perfect Agar Plates Every Time
1. Use a Recipe
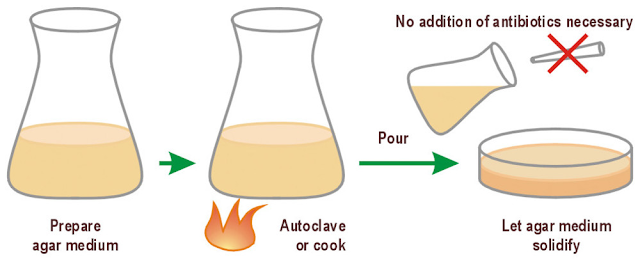
Make up the medium according to the recipe, then add the desired amount of agar (normally about 1% w/v) and stir. If you autoclave without stirring, with the agarose still floating on top of the liquid, you get an agarose cake in the medium. Interesting, but useless.
When making up the agar, only use 3/4 of the bottle’s volume. This allows space for bubbles to rise while the agar is melting in the microwave (and saves you cleaning up overflowing agar from the microwave!).
2. Autoclave
Autoclave your medium for 25 minutes. After autoclaving, you can, of course, store the medium-agar mix in a toughened glass bottle, then melt it in a microwave or water bath when needed. Make sure you use toughened glass bottles, or disaster can strike.
3. Cool It!
Cool the medium-agar mix to 55°C. For routinely consistent results, do the cooling for a couple of hours in a 55°C water bath. Agar starts to solidify at about 50°C. Using the water bath means you can consistently cool the mixture above the solidification temperature.
Before I used a water bath, I used to just cool it in the air but would inevitably forget about it and come back to find solidification had already started – lumpy plates are no good for spreading!
4. Supplement It
You can now add any antibiotics or supplements and be confident that the agar is at a suitable temperature because you have cooled it in the water bath.
5. Pour the Plates
Use about 30 mL of the agar-medium mix for each plate when using a 100 mm diameter plate. The less agar-medium mixture in each plate, the more quickly they dry out. 30 mL is a good amount for long-term storage; 10–20 mL is fine if you will use the plates relatively soon.
For consistency, I’d recommend using a serological pipette. Suck up 2-3 mL more than you need to minimize blowing bubbles into the plate.
6. Let It Set
If there are any bubbles in the plates, briefly pass the flame over to pop them. Classic error: trying to move the plates before they’ve set is just asking for trouble. Just leave them alone (and maybe admire your perfect agar plates while you wait)!
7. Get Dry
Dry the plates in the laminar flow hood with the lid slightly off for 30 minutes (or in a 37°C incubator for 2–3 hours, or room temperature for 2–3 days). Drying the plate is very important for storing the plates and growing colonies.
If you don’t dry the plates, the moisture will evaporate and condense on the lid during storage or incubation, giving you horrible wet plates. At worst, the humidity can affect the plating of your cells. Use a timer to remind you when the 30 minutes are up as – in my experience – it is straightforward to forget about your plates and come back to find your plates have turned into agar crisps/chips. Tasty.
8. Use It or Store It
Once you’ve poured your perfect agar plates, you can use them immediately or seal them for later use. You can use Parafilm or pop them in the bag that the plates came in for easy storage. Store the plates at 4°C. Guidelines suggest using agar plates within approximately 2 to 4 weeks.
Depending on the additives you have included, the shelf life of the prepared plates might be shorter – make sure you check this before you start so you don’t end up wasting your time (and resources) making too many plates.
A quick way to label your plates is to have a color code for each antibiotic and medium type you tend to use (e.g., red for ampicillin, black for kanamycin, green for LB, blue for M9). Stack the plates and use the appropriately colored lab marker to draw a line down the whole stack. Make sure you keep the color code to hand, though.
Now you should have perfect agar plates every time. If you’ve got any other ideas or additions to this protocol, please leave a comment.
For more protocols, please visit The phage protocols by clicking here
Phage work can be done using many different media depending on what is available to the researchers doing the experiment, although LB and TSB (A) are the most preferred.



Leave a Reply
You must be logged in to post a comment.