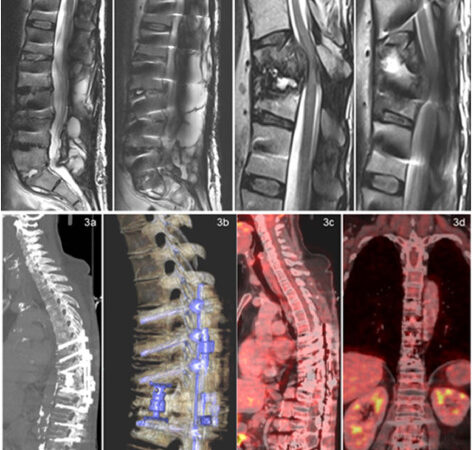
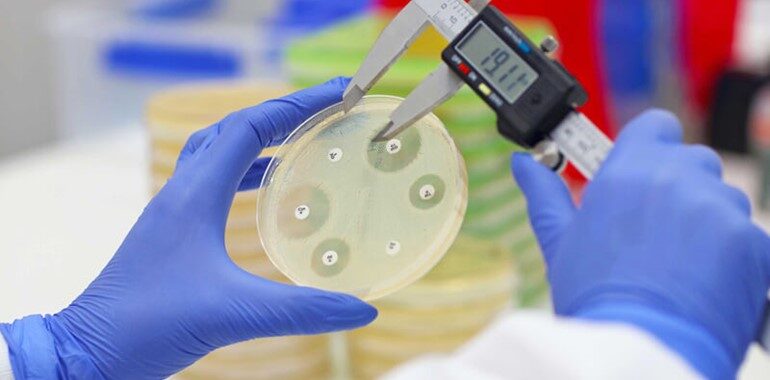
BiomX Inc. has announced the discontinuation of its Phase 2b clinical trial of BX004, a nebulized phage therapy under investigation for cystic fibrosis (CF) patients with chronic Pseudomonas aeruginosa infections. The decision follows an internal review and recommendations from the independent Data Monitoring Committee (DMC).
According to the company, the DMC conducted a safety assessment on November, 2025, during which unexpectedly high rates of adverse events were identified. After evaluating the committee’s feedback and performing its own internal analysis, BiomX elected to discontinue the trial.
Focus on Patient Safety and Program Feasibility
BiomX stated that patient health and safety were the primary considerations behind the decision. While alternative dosing or treatment strategies were considered, the company determined that the time, cost, and resources required to safely continue the program exceeded its current capabilities.
Jonathan Solomon, Chief Executive Officer of BiomX, emphasized the ongoing unmet needs within the cystic fibrosis community, particularly in managing chronic P. aeruginosa infections. However, he noted that continuing the BX004 program was not feasible given the company’s existing financial and operational constraints.
Strategic Refocus on BX011 and Cost Optimization
Following the discontinuation of BX004, BiomX plans to refocus its development efforts on BX011, a fixed phage cocktail targeting Staphylococcus aureus infections associated with diabetic foot infections (DFI).
About BiomX
BiomX is a clinical-stage biotechnology company developing natural and engineered bacteriophage therapies designed to target and eliminate specific pathogenic bacteria. Through its proprietary BOLT (BacteriOphage Lead to Treatment) platform, the company advances both fixed phage cocktails and personalized phage treatments aimed at addressing chronic and drug-resistant infections.
Read the complete news event here